Home » Uncategorized
Category Archives: Uncategorized
Chemoenzymatic Total Synthesis of ent-Oxycodone: Second‑, Third‑,and Fourth-Generation Strategies
J. Am. Chem. Soc. 2019, 141, 27, 10883-10904 full text link
文献汇报人:Hayatullah Imtiaz
汇报日期: 2019年12月14日
图文摘要:
-
推荐原因:
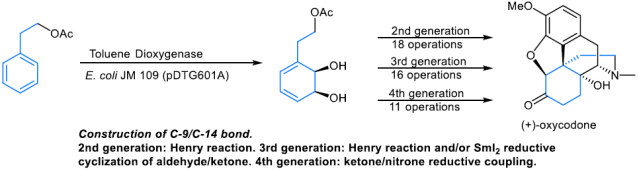
Four distinct approaches to ent-oxycodone were designed and accomplished. All rely on the same starting material, the diene diol derived from phenethyl acetate by the whole-cell fermentation with E. coli JM109 (pDTG601A), a strain that overexpresses toluene dioxygenase.
The key step in the first-generation approach involves the construction of the C-9/C-14 bond by a SmI2-mediated cyclization of a keto aldehyde. The second-generation design relies on the use of the Henry reaction to accomplish this task. In both of these syntheses, Parker’s cyclization was employed to construct the D-ring. The third generation synthesis provides an improvement over the second in that the nitrogen atom at C-9 is introduced by azidation of the C-9/C-10 olefin, followed by reduction and lactam formation between the C-9 amine and the Fukuyama-type lactone. Finally, the fourth generation takes advantage of the keto−nitrone reductive coupling to generate the C-9/C-14 linkage. The four generations of the total syntheses of ent-oxycodone were accomplished in 13, 18, 16, and 11 operations (19, 23, 24, and 18 steps), respectively.
A brief introduction to Ph.D Sergey V Pronin’s Recent works
J.Am.Chem.Soc.2015,137,15410 full text link
J. Am. Chem. Soc. 2016, 138, 12316−12319 full text link
J. Am. Chem. Soc. 2018, 140, 12770-12774
full text link
文献汇报人:谌雪鸣
汇报日期: 2019年7月6日
图文摘要:


-
推荐原因:
Sergey V. Pronin 小组近三年的工作对蕈青霉素类二萜化合物进行了系统地探究,成功使用HAT反应构筑了二萜核心骨架。并通过添加邻近基团地参与,调控了分子内的 aldol缩合反应,实现了立体选择性地改良,在2015年发表在JACS上。该组从反应前体合成中汲取经验,系统探究了烯醇硅醚与末端炔在InBr3催化下的偶联反应,在次年的JACS上发表。基于2015年的工作,其课题组对蕈青霉素衍生物Nodulisporic acids类化合物进行了合成研究。主要探索了π酸催化的炔烃与烯烃的合环,该反应一步构筑了两个环且收率高。这些工作关联性强且想法十分新颖。
Swinhoeisterols from the South China Sea Sponge Theonella swinhoei
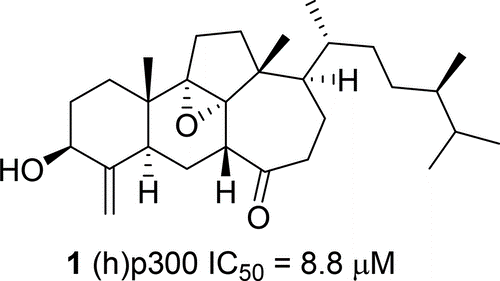
J. Nat. Prod., 2018, 81 (7), pp 1645–1650
DOI: 10.1021/acs.jnatprod.8b00281
Link: click
Swinhoeisterols C–F (1–4), four new steroids having a rearranged 6/6/5/7 ring system, were isolated from the Xisha sponge Theonella swinhoei, together with the known analogue swinhoeisterol A (5). Their structures were determined based on spectroscopic analysis, TDDFT-ECD and optical rotation calculations, and biogenetic correlations. In an in vitro assay, compound 1 showed an inhibitory effect on (h)p300 with an IC50 value of 8.8 μM, whereas compounds 2–4were not active.
